California Association of Criminalists

Since 1954

GLASS TECHNOLOGY AND PROPERTIES OF INTEREST TO THE CRIMINALIST
I.H. Malitson, G.W. Cleek, and C.P. Saylor, National Bureau of Standards, Washington, D.C. 20234
The Law Enforcement Standards Laboratory, National Bureau of Standards, has initiated a program to instruct criminalists in the characterization of glass. The program is sponsored by the National Institute of Law Enforcement and Criminal Justice, Law Enforcement Assistance Administration, Department of Justice.
A review of glass technology will describe types of glass, defects in glass, refractive and dispersive properties, heat treatment and density determinations (G.W. Cleek). A review of the microscopical determination of refractive index (RI) of glass fragments will cover measurements by immersion, control of immersion liquid, test of solid and liquid match, and calibration of comparison liquid. A homemade phase-contrast apparatus capable of 1 x 10-5 RI accuracy will be described (C.P. Sayior). The testing and use of the Abbe refractometer will be discussed, and the new NBS glass RI standard that has been developed for calibrating both refractometers and microscope immersion liquids will be described. Initial results of a study of the variation in RI of auto headlamp glass will be presented (I.H. Malitson). Audience participation in these discussions is invited.
QUALITY CONTROL: LAWS AND LOGIC
D.R. Morales, Clinical Chemistry Laboratory, California Department of Health, Berkeley
Some laws and regulations relating to laboratories are, becoming more specific and detailed with regard to day-to-day laboratory operations. Occasionally, there can develop resistance to newer laws and regulations based on the premise that they require scientists to engage in illogical acts. This presentation examines the proposition that there exists a disparity between laws and logic in the area of quality control in laboratories.
THE ORIGIN AND ACTIVITIES OF THE FORENSIC SCIENCE PROGRAMS AT THE GEORGE WASHINGTON UNIVERSITY
James E. Starrs, The George Washington University National Law Center, Washington, D.C.
Over the past five years, the George Washington University, in cooperation with various governmental institutions such as the F.B.I., the Armed Forces Institute of Pathology and local police agencies has developed an on and off campus degree program for persons interested in the affairs of forensic science. The uniqueness of this program stems from its interdisciplinary character combining curriculum features from law, science and business administration in which these various specialties within the G.W.U. have engaged in a cooperative effort. The program, now under the direction of the Department of Forensic Sciences at G.W.U., has also offered short courses for persons in the varied fields of the forensic sciences. Its future can be enhanced by now pending applications for federal financial support, although laboratory space and facilities have been, to an extent, budgeted by the G.W.U. for its use.
ANALYSIS OF PRIMER RESIDUES BY ATOMIC ABSORPTION SPECTROPHOTOMETRY
Edward F. Rhodes, School of Criminology, University of California, Berkeley, and Gary F. Gonzales, Orange County Sheriff's Department
The determination of barium and antimony levels from primer residues in gunshot cases has been the object of intensive research for over a decade. Until recently, the most effective methods have employed neutron activation analysis. The development of the carbon rod atomizer for the AA spectrophotometer, however, has brought this instrument's sensitivity within the limits of primer residue levels. It has also provided an instrument within the fiscal means of most laboratories. A series of tests were conducted using the Varian Techtron Model 1200 AA spectrophotometer with the Model 63 carbon rod atomizer. A number of methods for removing and extracting primer residues were tried, including cotton swabs, filter paper swabs, and PVA films. Different solvents were tested including 0.1% trisodium phosphate, several types of soap solutions, and 5% HNO3. The results indicated, superiority for some methods, brought out problems with the others and suggested some areas which merit further exploration and development. This note of our findings to date is offered in hopes of soliciting information and ideas from those labs which have attempted these, or similar, methods.
A VERSATILE SYSTEM OF CLOSE-UP PHOTOGRAPHY FOR CRIME SCENE OR LABORATORY
T.L. Coddington, San Francisco Police Department.Crime Laboratory, San Francisco
Hand-held close-up photography coupled with electronic flash has been found to be of great practical value in recording details of physical evidence often missed in routine crime scene photographs. The system is also useful inside the laboratory for recording the nature of physical evidence as submitted to the laboratory prior to beginning examination. A simple calibration routine is outlined to enable the photographer to determine exposures correctly. The same equipment can be used for photography at normal distances, and with minor attachments can be used into the region of photomicrography. Examples are given.
NON-RANDOMNESS OF STRIATION EVIDENCE
John I. Thornton and Vance Nordman, School of Criminology, University of California., Berkeley 94720
Consideration has been given to whether striation evidence may be considered to be deterministic or phenomenological in character. Various sub-classifications of deterministic data (periodic, non-periodic, sinusoidal, transient, etc.) and various sub-classifications of random data (stationary, non-stationary, ergodic, non-ergodic, etc.) are reviewed and the randomness of toolmark and bullet evidence is tested by means of the following measure of randomness in a series of alternatives:
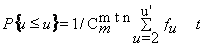 | m = number of events n = number of non-events u = number of groups formed |
For high m and n, the calculation is quite tedious. A FORTRAN program was written which has resulted in a table extended to n of several thousand. In working with actual toolmarks, the width of the narrowest resolvable striation is taken as an event, and groups are formed as runs of this dimension. In all of the toolmarks and bullets examined thus far, the probability of randomness is < 0.05.
Frieda S. Swed and C. Elsenhart. Tables for testing randomness of grouping in a sequence of alternatives. Annals of Math. Stat. 14: 66 (1943).
A STUDY OF GUNSHOT RESIDUE PATTERNS ON TARGETS VIA THE RHODIZONATE TEST FOR LEAD
Jan S. Bashinski, Criminalistics Section, Oakland Police Department
Gunshot residue patterns were prepared representing a number of variables, including type and caliber of weapon, type of ammunition, and muzzle to muzzle distance. The gunshot residue patterns on these test targets were developed with the sodium rhodizonate procedure (CAC Spring 1974) which reveals the distribution of lead residues. These experiments provided a basis for assessing the relative contributions of the bullet alloy material and the primer material to the total lead residue pattern for jacketed versus unjacketed bullets. Data from tests on a .38 cal revolver was used for the construction of a three dimensional "Model" of a gunshot residue pattern, depicting the lateral as well as the forward distribution of lead residues during the firing of the weapon. Corollary experimental work included preliminary evaluation of the relative merits of the Walker nitrite test, soft X-ray photography, and the sodium rhodizonate test in revealing a gunshot residue pattern.
Some comment will be made on the implications of this work relative to the interpretation of results obtained on targets and hand washings by NAA or other instrumental techniques - emphasizing the need to guard against contamination in test firings and to consider the effect of all variables when attempting to interpret a residue pattern in a case situation.
RELIABLE TRACE GAS ANALYSIS SYSTEMS (TGAS) FOR ATMOSPHERIC CONTAMINATION
M.L. Moberg, Analytical Research Laboratories, Inc., Monrovia
For more than 10 years and over 1000 gas analyses for atmospheric contaminants (1 ppm to 1 ppt) ARLI has found that 3 major factors contribute to make (TGAS) meaningful and reliable. These are: (1) type and representation of sampling, (2) clean and reproducible blank or empty sampling containers with self-imposed quality controls, and (3) substantial reduction or elimination of "ghosting" or trace component adsorption in analytical instrumentation. By carefully adhering to good analytical practices incorporating these factors and combining them with good instrumentation, high confidence level data production can be expected. Optimized instrumentation can include: electronic integration, high efficiency gas chromatography (2 columns & temperature programming) with multiple detectors and if possible mass spectrometry tandemized with the chromatograph for unknown peak identification.
RAPE AND THE UTILIZATION OF PHYSICAL EVIDENCE IN ITS INVESTIGATION AND PROSEUCTION
George Sensabaugh, University of California (Moderator), Two representatives from BAWAR (Bay Area Woman Against Rape), Clayton DaVega, Alameda County District Attorney's Office, Harvey Homel Alameda County public Defender's Office, Thomas Johnson, Berkeley Police Department, Nancy Rockeman, Legislative Consultant to Assemblyman Kenneth Meade; Arlene West, Attorney, E.T. Blake, University of California, R.P. Erickson, Department of Pediatrics, University of California, San Francisco, Leonard Ortega, Institute of Forensic Sciences, Oakland
The crime of rape has become a focus of increasing social concern. One reason for this concern is the increase in volume of reported forcible rape offences; the Uniform Crime Reports for 1973 show an increase of 62% since 1968, and it is estimated that a substantial proportion of rape offences remain unreported. Another cause for this concern is that only about half of the reported rapes are cleared by an arrest and of these, about 65% are not prosecutable or upon prosecution result in acquittals or dismissals; these figures are suggestive of major problems in rape investigation and prosecution. Finally, there has developed in the past few years an increasing awareness of and sensitivity to the plight of the rape victim. This has resulted in substantial modifications in police and hospital procedures in dealing with rape complaints. It has also led to the enactment of legislation modifying judicial procedures with respect to the prosecution of rape.
In this symposium, the problem of rape, its investigation, and its prosecution will be considered in two contexts. The first half of the symposium will consider factors and concerns attendant to the utilization of physical evidence in rape investigation. The second half will be concerned with physical evidence itself, specifically focusing upon sperm and semen and their value as evidence materials.
DISCUSSION OF FACTORS RELEVANT TO THE UTILIZATION OF PHYSICAL EVIDENCE IN RAPE INVESTIGATIONS
THE ROLE AND INFORMATION CONTENT OF PHYSICAL EVIDENCE IN RAPE INVESTIGATION
LOW TEMPERATURE PHOSPHORESCENCE AS AN ADJUNCT TO THE ACID PHOSPHATASE TEST FOR THE IDENTIFICATION OF SEMINAL FLUID
P.F. Jones, A.R. Calloway, D.J. Carre, and S. Siegel, The Aerospace Corporation, El Segundo
For the identification of seminal fluid, the acid phosphatase test has received the most attention in criminalistics laboratories. However, due to considerable controversy pertaining to the specificity of this method, another more specific method is required. Low-temperature phosphorescence has been demonstrated to be a suitable method of locating suspected seminal stains on cloth. Additional tests have demonstrated that the low-temperature phosphorescence can also be employed to differentiate between seminal fluid and other materials known to give positive acid phosphatase test results. Only two materials tested, vaginal fluid and rattlesnake venom, gave phosphorescence results that were difficult to separate from that of seminal fluid, and the rattlesnake venom was easily distinguished using other luminescence characteristics. In this respect the low-temperature phosphorescence can be used as a valuable adjunct to the acid phosphatase test for the identification of seminal fluid.
BARBITURATE CHARACTERIZATION BY LINEAR OVEN PROGRAMMED G.L.C. ON OV-1 AND OV-17 COLUMNS
Allen J. Boudreau, Criminalistics Laboratory, Fresno County Sheriff's Department, Fresno
Current specimens of "street reds" frequently have been found to contain a mixture of two or more barbiturates. Speculation on the possible reasons for this occurrence can lead in several directions such as: the mixture of different barbiturates; the use of a mixture of different substituted malonic acids; a lack of aptitude of the synthesizer of the substituted malonic acids; et infinitum ad nauseum, Numerous G.L.C. experimental conditions for the separation of barbituric acid derivatives have been published. However, experiments with twenty-five barbiturates available have made the fact apparent that under a number of experimental conditions certain barbiturates are unresolved or the re-solution is satisfactory but at the expense of long retention times. A study was initiated to determine which barbiturates would be unresolved under certain experimental conditions and what experimental conditions would result in adequate resolution with reasonable retention times. The utilization of Linear Oven Programming is compared to Isothermal operation and is shown to have value. Data is presented for six foot glass OV-1 and OV-17 columns.
A RAPID METHOD TO IDENTIFY BARBITURATES BY INFRARED SPECTROSCOPY
A.J. Boudreau, Criminalistics Laboratory, Fresno County Sheriff's Department, Fresno
Barbiturate differentiation by Infrared Spectroscopy requires a comparison of the unknown spectrogram to a standard spectrogram. Due to the variety of barbituric acid derivatives manufactured a time consuming search of standard spectrograms may be necessary to locate the standard spectrograms for the unknown. A quick check of the unknown spectrogram to locate the wavelengths (cm-1) of the four strongest absorption bands in the 1200 cm-1 to 400 cm-1 region, followed by reference to a chart, will indicate which standard spectrogram to consult for the comparison. Experimental methodology; absorption band selection parameters; a chart of the four strongest absorption bands; and Infrared spectrograms for twenty-five barbiturates are presented. The barbiturates include both common and infrequently seen derivatives. Handouts will be provided.
A NEW SELF-SEALING PRESERVIAL FOR BREATH ALCOHOL ANALYSIS
Naresh C. Jain, USC School of Medicine, Los Angeles, and Rancho Los Amigos Hospital, Downey & Wallace Van Note, Omicron Systems Corp., Palo Alto
In People v. Hitch (Crim. 16915), Super. Ct. No. APP 180, April 10, 1974), Supreme Court of California upheld the trial court's decision that the prosecuting agencies should preserve the breath analyzer ampoule in accordance with due process which the defendant now has with blood and urine. On its own motion, however, the Supreme Court granted a rehearing which, at the time of this writing, had not taken place. As a result of Hitch ruling, we describe herein an inexpensive, simple to use, self-sealing PRESERVIAL for use in breath analyzer. The system consists of an optically clear vial prefilled with a known quantity of acid/di-chromate/AgNO3, solution. The PRESERVIAL may include a volumetric scale. The PRESERVIAL is closed with a self-sealing Teflon-backed rubber septum and sealed with a crimp seal or screw cap. Breath sample is introduced through a specially designed Teflon-coated needle (scrubber) and cannula (exhaust). The needle/cannula assembly is inserted through the septum so that the needle is below the surface of acid/dichromate solution, and the cannula is located above the test medium. This allows a continuous flow of breath. Following analysis, needle/cannula assembly is removed, allowing the stopper or septum to self-seal, and, thus, maintain the reactant materials in an air-tight condition. Test data show no change occurring in the unused PRESERVIAL. Further study is in progress to show the preservation of the post analysis PRESERVIAL. OMICRON Systems Corp. (Patent Pending).
CO-SWEEP PRESERVIAL FOR CAPTURING ALCOHOL AND OTHER VOLATILES FROM BREATH
Naresh C. Jain, USC School of Medicine, Los Angeles and Rancho Los Amigos Hospital, Downey & Wallace Van Note, Omicron Systems Corporation, Palo Alto
A simple system for the collection, concentration and preservation of volatile intoxicants such as alcohol, acetone, and possibly cannabinoids from breath is described. The system gives due process in providing a sample of alcohol in breath to defendants charged with drunken driving. The collection device is a small portable unit or accessory which is easy to operate. The system operates on the principle of co-sweeping volatile substances from the vapor (breath) into a small volume of liquid having greater affinity for the volatile compounds. The CO-SHEEP PRESERVIAL employs a needle/cannula concept for introducing the sample into a liquid absorbing medium. The liquid is contained in a micro conical, rectangular or other shaped vial which is crimp sealed or screw capped by a Teflon-lined, inert or nonreactive septum.
For GC analysis, the CO-SWEEP PRESERVIAL may contain or appropriate internal standard with a preservative in an aqueous medium in which alcohol is absorbed. Similarly, PRESERVIALS of varied shapes and sizes are designed for direct use in a spectrophotometer or for analysis using different techniques. The absorbing liquid could be colorless or colored, reactive or non-reactive which, if desired, may produce a specific color or reaction with the compound of interest in the breath. Extension of this principle is made to test acetone in the breath of diabetics, industrial solvents in workers, and cannabinoids in marijuana smokers, volatile solvents in glue snifters and for similar applications in industry. OMICRON Systems Corp. (Patent Pending)
STUDIES ON A REPLACEMENT FOR THE BENZIDINE TEST AS A PRESUMPTIVE TEST FOR BLOOD
M.A. Blake, Criminalistics Laboratory, Contra Costa County Sheriff's Office, Martinez
Benzidine has recently been recognized as a carcinogen and many laboratories have had to discontinue its use. This has necessitated. research to provide a suitable replacement with regards to sensitivity, specificity, and convenience. Four reagents were studied:
These studies indicate that the modified phenolphthalein test is a suitable replacement for the benzidine test.
DENSITY DETERMINATION ON POLYMERS AMD POSSIBLY OTHER TRACE EVIDENCE
John I. Thornton, School of Criminology, University of California, Berkeley 94720
A method is described for the determination of density based upon volume displacement. This is an application of Archimedes' principle, and certainly no claim for novelty is being asserted. An old, broken microburet is sealed at the broken end and partially filled with dry Portland cement. The volume is noted. Fragments of polymer are weighed as care-fully as possible, pushed under the surface of the cement in the buret, and the volume is again noted. (The volume may be read to 0.01 ml). The weight per unit volume (density) can then be calculated. Precision is achieved only if the buret of cement is tamped by dropping lightly on a tabletop 50 times. The reproducibility is on the order of + 2%. The particle size of the cement is so fine that it acts as a liquid, filling the voids around irregularly shaped particles. The technique has some advantages over water displacement in some instances; certain polymers (polyethylene and polypropylene) are lighter than water, and water cannot be used. With other polymers, surface tension presents some problems. Some polymers are adversely affected by non-polar liquids, barring the use of these liquids even though their density is less than that of polyethylene and polypropylene. For materials which are soluble, float, or are otherwise affected by true liquids, the cement technique could be used. The technique seems to hold promise for other types of evidence.
ELEMENT PROFILES OF PAINT CHIPS BY X-RAY FLUORESCENCE SPECTROMETRY
Lucien C. Haag, Criminalist, Phoenix Police Crime Laboratory, Phoenix, Arizona
Paint chips ranging in size down to 2 mm are analyzed with the EDAX energy dispersive X-ray spectrometer system for all elements above Ne. Elements present in the sample are ranked in descending order on the basis of peak height with the major peak taken as 100. The analysis is entirely non-destructive, requires no sample preparation, and is routinely completed in 30 seconds to 8 minutes on typical paint chips. Precision and sensitivity are sufficient to detect differences between paint chips from different areas of the same vehicle. Approximately 100 automotive paint samples have been examined to date with no recurrence of element profiles between samples of different origin.
CHARACTERIZATION OF BROWN HEROIN BY ELEMEMTAL ANALYSIS
L.J. Berens, Drug Enforcement Administration Laboratory, Dallas, Texas & M.D. Miller, Drug Enforcement Administration Laboratory, San Diego
A drug library of more than 200 brown heroin exhibits representing major seizures by the Drug Enforcement Administration in the Arizona and Texas areas over the past two years were examined for similarities in trace element content by Atomic Absorption Analysis (AAA) and Energy Dispersive X-Ray Analysis (EDX). The immediate objective of this study was to characterize these brown heroin specimens through elemental profiles and determining whether information useful for enforcement intelligence had been developed. The brown heroin preparations were found to possess unique elemental compositions which appeared to vary in concentration as a function of the heroin potency. Six Indices of comparison were utilized for ease examinations on a localized basis; they were (1) heroin, (2) procaine, (3) Ca, (4) Zn, (5) Fe, and (6) Mg content. Comparative case analysis was based on the determination of heroin and element content, procaine to heroin ratios, selected elemental ratios, and any other supporting evidence such as EDX spectra or the presence of water soluble dyes. Atomic Absorption determinations were made on the ele-ments Ca, Zn, Fe, and Mg which were found in widely varying concentrations in all samples examined. The authors feel at the present time that the elemental composition which typifies brown heroin is dictated to a great degree by the process of synthesis which is often very crude in scope.
ESTERASE D POLYMORPHISM IN BLOOD STAINS
Edward T. Blake and George F. Sensabaugh, School of Criminology, University of California, Berkeley
A new genetic polymorphism, Esterase D (EsD), has been recently described by Hopkinson et al (Ann. Hum. Gen. Lond... 37 (1973), 119-137.) The existence of two common alleles makes this enzyme a useful genetic marker for the typing of blood stains. An additional attractive feature of this system is that Esterase D can be typed concurrently with EAP or PGM on the same gel surface. The substrate for Esterase D, 4-methylumbelliferyl acetate, is of limited commercial availability but is easily synthesized in good yield and purity from 4-methylumbelliferone and acetic anhydride. Procedures for synthesis and data concerning the stability of this substrate will be described. Information on the persistence of Esterase D in blood stains will be presented.